ABSTRACT
Destruction of calcified tissue was caused by acids which are by product of carbohydrate metabolism of acidogenic bacteria consequent to dental caries. The purpose of this study was to assess the existence of acidogens potentially causing the dental caries and comparatively evaluation of efficacy of different toothpastes. The dental plaques of fifty persons belong to three age groups (1 - 20, 21 - 40, 41 - 60 year and above) were examined to identify microorganisms by the culture method. Thirty nine bacteria were isolated by spread plate method on BSMY I minimal media. Thirteen out of thirty nine, acidogens colonized in the dental plaques. Seven potentially acidogens CD17, CD26, CD27, CD28, CD29, CD34 and CD35 were treated with five different toothpastes. Inhibition effect of Triclosan and Fluoride containing tooth pastes were found more efficient. The results of the present study revealed that bacteria that commonly cause dental caries colonized in dental plaques of children and alcoholic person. Therefore, dental plaques must be considered a specific reservoir of colonization and subsequent dental caries. To reduce the dental problem triclosan and fluoride containing product should be recommended.
1. Introduction
Dental caries is one of the most common chronic infectious diseases in the world [1,2]. Bacterial plaque accumulated on dental surfaces and composed of native oral flora is the primary etiologic agent of dental caries. Cariogenic bacteria interact by various recognized ways including co-aggregation [3], metabolic exchange, cellcell communication [4], and exchange of genetic material [5]. These mechanisms benefit bacterial survival and can make dental biofilms difficult therapeutic targets in dental diseases. Dental caries cause destruction of enamel, dentin or cementum of teeth due to bacterial activities. The burden of dental caries is still a major health problem in most industrialized countries as it affect 60% - 90% of school-aged children and the vast majority of adult. This is largely due to the increasing consumption of sugar and inadequate exposure to fluorides [6].
Triclosan, a chlorophenol derivative, kill germs by interfering with the enzymes required for fatty acid synthesis. Similar to triclosan the fluorinated products were also found to possess marked antibacterial activities. These active compounds were reducing cariogenic bacteria to strengthen the teeth by reducing demineralization and increasing remineralization of teeth [7].
Biofilms are surface-adherent population of microorganisms consisting of cells, water and extra cellular matrix material [8]. Streptococcus mutans, the principle cariogen for dental caries, co-exist with over 500 other species of bacteria as an interactive community known as the dental biofilm [9]. A quorum sensing signaling system is essential for genetic competence to function optimally in Biofilms [10]. Dental caries is a biofilm-dependent oral disease, and fermentable dietary carbohydrates are the key environmental factors involved in its initiation and development. Dental caries results from the interaction of specific bacteria with constituents of the diet within a biofilm termed “dental plaque” [11]. Sucrose is considered the most cariogenic dietary carbohydrate, because it is fermentable, and also serves as a substrate for the synthesis of extracellular (EPS) and intracellular (IPS) polysaccharides in dental plaque [11,12]. Enamel integrity is disrupted secondary to the formation of a dental biofilm and the caries process occurs along the interface between the dental biofilm and the enamel surface [13,14]. Cariogenicity of sucrose has been associated with its frequency of exposure and concentration [15,16].
In order to understand and to know detail information on the micro flora of dental plaque in the individuals having different food habit and brush timing of different age groups, we have designed microbiological study, with special attention to the presence or absence of acidogenic bacteria in the dental plaque stored in reduced Transfer media. The purpose of this study was to assess the prevalence of dental plaque colonization by potential acidogens and to compare the efficacy of commercially available tooth pastes against those acidogens.
2. Material and Methods
2.1. Sample Collection
Fifty patients of five different zone of Bilaspur (C. G.) participated in this study. Ten samples from each zone were collected. However, food habit was similar for all, only differences were the brush timing and age. Three age groups were 1 - 20 (I), 21 - 40 (II), 41 - 60 and above (III) years. I group consume more sugar stuffs, II group consume all food stuffs in optimal amount but III consume alcoholic beverages and also have low immunity. Dental plaques from all patients were picked up through forceps and transferred into 2 ml of reduced transport fluid medium (0.4% agar, 0.15% thioglycollate/phosphate buffered saline) [17] in sterile tubes and stored at 4˚C and processed within one night of collection [18].
dental laser handpiece After explanation of the purpose of the study, all subjects or their families voluntarily provided informed consent in writing.
2.2. Growth Media for Bacteria Isolation
Basal salt medium with yeast extract (BSMY I) were used throughout this study. BSMY I [19], used for the isolation of bacteria from dental plaque samples contained: 1.0 g yeast extract, 0.3 g (NH4)2SO4, 0.14 g MgSO4·7H2O, 0.2 g CaCl2·2H2O, 0.1 g NaCl, 0.05 g KH2PO4, 0.05 g K2HPO4, 0.6 mg H3BO3, 0.17 mg CoCl2·6H2O, 0.09 mg CuCl2·2H2O, 0.1 mg MnCl2·4H2O, 0.22 mg ZnCl2, 10 g glucose in 1 liter of Tris-HCl buffer (pH 8.0). All the chemicals were used of analytical grade.
2.3. Isolation of Bacteria from Dental Plaque
Each sample in tubes was inoculated separately into 25 ml of BSMY I broth. Inoculated flasks were incubated at 35˚C ± 1˚C for 48 hours. Freshly grown culture of 1 ml from each dental plaque was serially diluted up to 10−5 with distilled water. 100 µl serially diluted samples were spread over BSMY I agar plates. The inoculated plates were incubated at 37˚C for 3 days under aerobic conditions. The isolated colonies were picked up and subsequent streaked on slant of BSMY I for pure culture preservation [20].
2.4. Identification of Bacteria
2.4.1. Cultural Characteristics of Acidogens
Cultural characteristics of isolated bacteria such as size, shape, pigmentation, elevation and margin of colony were recorded by culturing them on BSMY I medium and incubated for 24 - 48 hours at 37˚C ± 1˚C. The colonies were observed under transmitted and reflected light condition to understand their optical properties. For example colony characteristics were considered for CD26 was 2 - 4 mm diameter, irregular, white, convex/flat, raised, smooth, translucent colony under light [21].
2.4.2. Morphological Characteristics
The bacteria were gram stained and observed under light microscope (40×). The shapes of bacterial cells were coccid, bacilli, single, paired, chain and dense clusters. CD26, CD28 are coccids and gram positive [20].
2.4.3. Biochemical Properties
Biochemical properties of the isolates were tested according to Bergey’s Manual of Systematic Bacteriology [22]. The following properties were determined: Catalase test,
dental laser handpieceacid production from carbohydrates glucose.
2.4.4. Antibiotic Susceptible Test
The susceptibility of the bacterial isolates to antimicrobial agents was determined using disk diffusion method [23] and interpreted according to Clinical and Laboratory Standards Institute (CLSI) [24] manuals. The following antimicrobial agents were obtained as standard reference disks for their known potency in laboratory use: Vancomycin (V) 10 μg, erythromycin (E) 15 μg, chloramphenicol (C) 10 μg (Himedia). All of these tests were performed on plates of BSMY I. A 0.5 ml of suspension of tested bacterial isolates was applied BSMY I plates. Antimicrobial disks were placed on the agar with sterile forceps. The agar plates were incubated inverted at 35˚C for 24 hours. Results were recorded by measuring the inhibition zone.
2.4.5. Toothpastes Assesses with Antibacterial Activity
Five tooth pastes were used in the present study. Effects of Toothpastes were based on content they possess (Table 3). Tooth pastes stock was prepared in sterile distilled water (0.5 g/ml). Acidogens were seeded to minimal media and made 3 mm well with the help of sterile cup borer. The samples were loaded (25 μl) in a well and plates were kept in incubator at 35˚C ± 2˚C for 24 hrs [25].
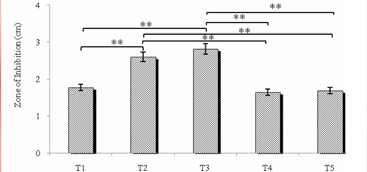 3. Result and Discussion
3.1. Prevalence of Bacteria
3. Result and Discussion
3.1. Prevalence of Bacteria
From fifty samples of dental plaques thirty nine bacteria were isolated abbreviated as CD1 to CD39. Harshal [26] has reported as dental caries with a prevalence of as high as 60% - 80% in children. Near 80% of the opportunistic bacteria isolated from plaque were found in saliva or tonsils, while 63% - 64% of the microbes present in saliva were present in dental plaques or tonsils [18]. This indicates organisms might be released from tooth surfaces into saliva and then colonizes on oral cavity surfaces such as the tonsils.
3.2. Acidogens
On the basis of acid production 13 bacteria were acidogenic. The potentially 7 acidogenic bacteria were subsequent treated with antibiotics and toothpastes, details are available in Table 1. Svensater et al. [27] had reported that nature of bacteria ability to produce acid (acidogenic) and tolerate acid environment (aciduric) are important that enable them to exhibit high dental caries. In India, a survey was carried out on school children showed caries prevalence of approximately 58%. Surveys among the U.S. population showed an incidence of 45.3% in children and 93.8% in adults [28].
dental laser handpieceThe enormous amounts of money and time are spent in treating dental caries. Hence, the prevention and control of dental caries is the main aim of public health, eventually the ultimate objective of public health is the elimination of the disease itself.
3.3. Antibiotics Sensitivity Test
The result of disk diffusion method revealed that most acidogenic bacteria were found resistant to antibiotics tested (Table 2). Out of seven acidogenic bacteria four were resistant to vancomycin, chloramphenicol, and erythromycin. Maripandi et al. [7] similarly reported that dental caries bacteria were resistant against vancomycin, chloramphenicol, penicillin, bacitracin and streptomycin.

